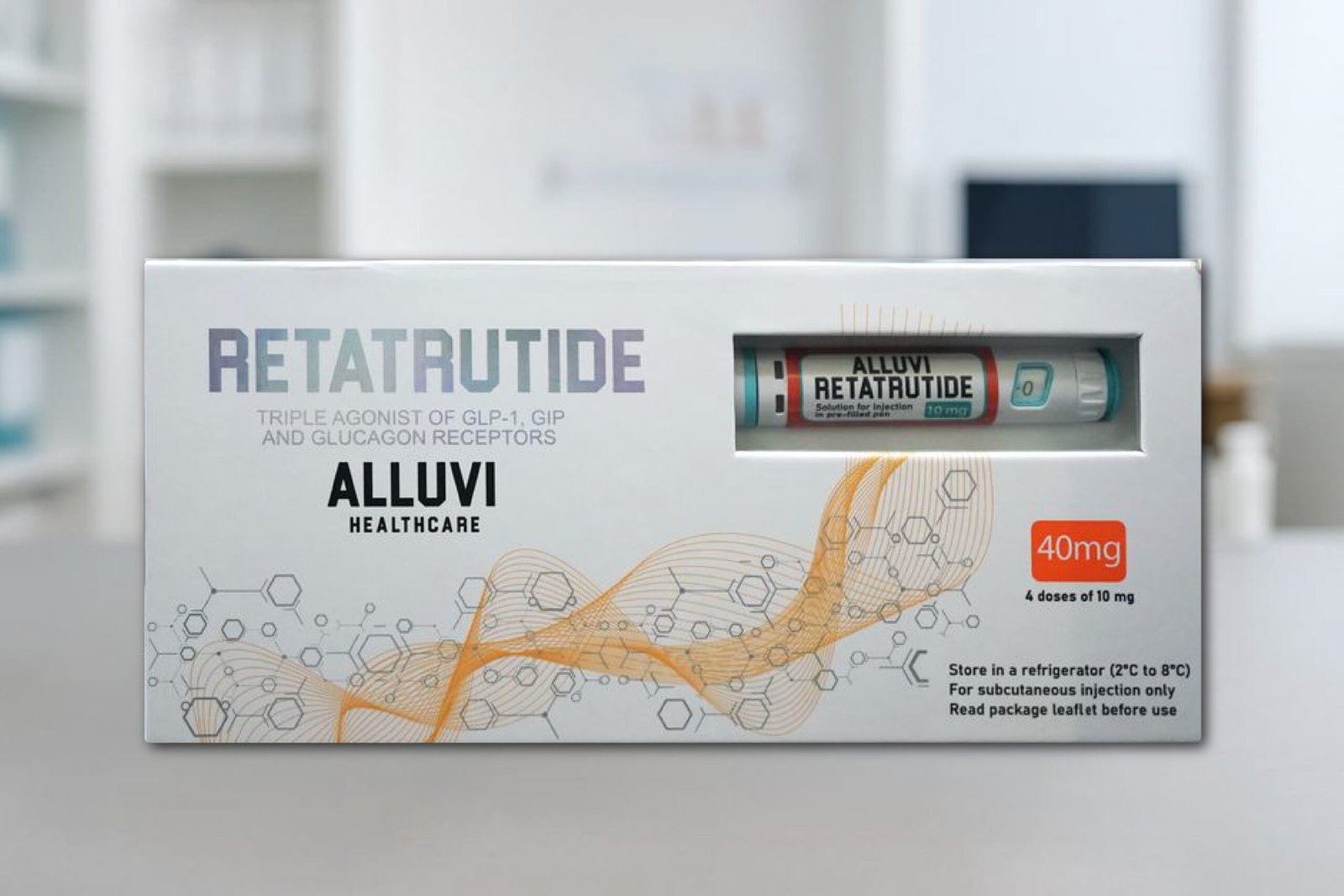
Alluvi Retatrutide 40mg
£170.00
Alluvi Retatrutide 40mg Pen, Alluvi Retatrutide 40mg. The novel experimental peptide retatrutide, which is now being studied for its potential in the treatment of obesity, diabetes, and metabolic health, is administered using the Alluvi Retatrutide 40mg Pen, a research-grade device.
Alluvi Retatrutide 40mg Pen | Alluvi Retatrutide 40mg
Alluvi Retatrutide 40mg Pen, Alluvi Retatrutide 40mg. The novel experimental peptide retatrutide, which is now being studied for its potential in the treatment of obesity, diabetes, and metabolic health, is administered using the Alluvi Retatrutide 40mg Pen, a research-grade device. Retatrutide is unique in that it functions as a triple agonist, stimulating glucagon receptors, GIP (glucose-dependent insulinotropic polypeptide), and GLP-1 (glucagon-like peptide-1). Due to its multi-receptor action, retatrutide is one of the most innovative drugs being studied in metabolic research. The Alluvi 40mg formulation’s pre-filled pen version is not meant for consumer or clinical usage; it is only meant for laboratory research and development (R&D).
Alluvi Retatrutide 40mg Pen | Key Features
- High-Dose Formulation: This sealed, ready-to-use pen contains 40 mg of retatrutide.
- Research Use Only: Not authorized for medical administration; strictly meant for laboratory research.
- Storage Needs: To preserve compound stability, it must be refrigerated between 2 and 8°C; freezing is not advised.
- Packaging: Provided in a sealed container with cold packs to maintain integrity while being transported.
- Contents: Usually comes with a study information page and a pen for safe handling.
Alluvi Retatrutide 40mg Pen | Research Applications
The main purposes of the Alluvi Retatrutide 40 mg Pen in scientific settings are:
- Examine the stability of compounds under various handling and storage circumstances.
- Examine how high-dose triple-agonist peptides are delivered.
- Perform formulation experiments that could guide future clinical applications in the study of obesity and metabolism.
Retatrutide’s Clinical Context
Because of its triple receptor action, retatrutide has demonstrated tremendous potential in clinical trials. Targeting GLP-1, GIP, and glucagon receptors concurrently has shown promise for:
- Boost type 2 diabetes’s glucose control.
- drive significant weight loss in tests on obesity, frequently surpassing the effects of dual agonists such as tirzepatide.
- improve metabolic control, providing more extensive therapeutic advantages than single or dual agonist substances.
Nevertheless, the Alluvi Retatrutide 40 mg Pen is not a medicinal product that has been authorized. Rather than being used directly on patients, this research-grade formulation is intended to facilitate laboratory exploration.
Aspects to Consider
- Not for Human or Veterinary Use: This product is only used for research and development, even if it is advertised online.
- Safety Procedures: To maintain the effectiveness of compounds, proper handling and storage are crucial.
- Potential Impact: More advanced peptide therapies may be developed with the aid of this technique in future studies.
Be the first to review “Alluvi Retatrutide 40mg” Cancel reply
Related products
Alluvi Retatrutide
Alluvi Retatrutide
Alluvi Retatrutide
Alluvi Retatrutide
Alluvi Retatrutide
Alluvi Retatrutide
Alluvi Retatrutide
Alluvi Retatrutide







Reviews
There are no reviews yet.